Cruel and unnecessary deaths
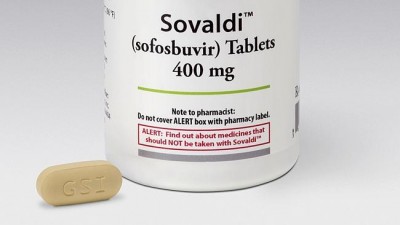
Last year 630 Australians died as a result of Hepatitis C. More will die, reports Graham Osborne, while the Pharmaceutical Benefits Advisory Committee rejects calls to make treatment affordable.
Australians are dying cruel and unnecessary deaths since a federal advisory board rejected funding for breakthrough Hepatitis C (HCV) treatment, say health experts.
The new drug Sofosbuvir, marketed as Sovaldi, has shown an unprecedented 90-95 percent success rate in curing HCV patients when used in combination with other available drugs.
Pharmaceutical giant Gilead Sciences has priced its “transformational” medicine at US$1,000 a pill or $US84,000 for a 12-week treatment. The company posted a record US$10.3 billion worth of sales of the drug in 2014 – its first year on the market.
The Therapeutic Goods Administration has approved Sofosbuvir for use in Australia but the Pharmaceutical Benefits Advisory Committee (PBAC), an independent body appointed by the Federal Government, last year refused to subsidise the treatment, citing an “unacceptably … high financial impact on the health budget.”
Gilead says in a statement that it’s now negotiating with the committee and remains “optimistic” that the clinical benefits and cost-effectiveness of Sofosbuvir will be recognised at the PBAC’s next meeting.
The PBAC has refused to comment on the negotiations.
Australia is one of the few developed countries in the world that has declined to subsidise Sofosbuvir for the most seriously ill HCV patients.
“People are dying and others are getting sicker and sicker,” says Dr Miriam Levy, the director of gastroenterology at Sydney’s Liverpool Hospital.
“There’s a lot of people at the edge of the cliff and you really can’t predict which ones are going to fall off.
“It’s terribly cruel and completely unacceptable not to treat these patients when there’s a drug that could cure them,” she says.
The Federal Government should follow the lead of the United Kingdom and allow immediate access to the new treatment for urgent cases, says Dr Levy.
“In the UK they’ve said, absolutely correctly, that while they’re negotiating for a better price the people with cirrhosis cannot afford to wait, so a special budgetary allocation was made to allow treatment for clinically urgent cases.
“If someone would just give me 30 treatment courses now at Liverpool Hospital. Let me deal with the sickest 30. Then we could meet again in six months and think about broader access,” she says.
Many HCV patients don’t realise that they’re being denied potentially life-saving treatment, says Dr Levy.
“One of my patients is an army veteran who fought on the battlefields for Australia. He has serious complications from cirrhosis and his lungs are shutting down and he has no idea that he’s missing out on something that could save his life. It’s just not right. This man fought for his country.
“I didn’t train as a palliative care physician and that’s not my brief, to support people in dying. But I’m being forced, as their liver specialist, to provide best supportive care to people because we’re living in a country where treatments are not available,” she says.
An estimated 233,000 Australians have chronic HCV infection, according to the latest figures from medical researcher the Kirby Institute. Of those, 80,000 have moderate to severe liver disease and more than 600 people die every year.
“The only way we can stop this epidemic is to get broad access to these new treatments for every Australian with the virus,” says Stuart Loveday, CEO of health lobby Hepatitis NSW.
“It’s a shocking, life-threatening situation and, certainly, unnecessary deaths are happening.
“We are on the cusp of a treatment revolution and we could virtually eliminate this blood-born virus from Australia within a generation but we need the necessary tools right now to allow equal treatment access,” he says.
It would be “much more cost-effective” to treat HCV in the short term rather than to deal with significant increases in cirrhosis, liver cancer, liver transplants and deaths that are expected in the next 15 years without the new treatments, says Loveday.
“Gilead did give 150 places for free for people on the liver transplant list but many others have been left waiting and they’re fearful that their liver disease will worsen and the delay could be months or even years,” he says.
Patients are being driven to the brink and some are coping very badly, says Loveday.
“We’re hearing from an agitated and angry community of people who are growing increasingly desperate.
“They’re looking at their options and some are considering travelling overseas to get the drugs at reduced prices or buying cheap drugs online,” he says.
Dr Levy has warned patients about the dangers of online ads offering cheap Sofosbuvir treatments.
“The quality control could not be guaranteed and they could end up buying lolly water made in backdoor factories,” she says.
Australia’s health system is good enough that we should not be forcing people into these kinds of desperate measures, says Loveday.
“We’re extremely disappointed that people living with Hep C in Australia cannot access these new therapies and we think it’s unethical to make them wait,” he says.
Loveday has called on the Federal Government and Gilead to immediately resolve the impasse and says “the best chance for a leap forward” will be at the PBAC meeting next month.
“The pharmaceutical industry is a little good/slash/evil and we just need them to be a little less greedy,” says Gren, a 60-year-old mental health researcher from Sydney who was first diagnosed with Hepatitis C 25 years ago.
Gren has undergone two treatments of the controversial HCV drug Interferon without success.
“The second Interferon treatment had so many spectacularly varied and terrible side effects that it left me unable to work for 18 months and now the virus that I can’t seem to shake is causing more and more problems in my life.
“These new treatments are the most exciting thing that has happened since I was diagnosed and they really are a revolution, a game-changer,” he says.
But Gilead needs to go away and make a “cost adjustment,” says Gren.
“I recognise their right to make a profit and to return research costs and all that but, I mean, how much profit? They’ve made record profits out of this, billions and billions of dollars.
“Perhaps it’s time to reduce the price and alleviate people’s suffering.
“I do want the government to contain costs and I do want them to negotiate on price but, on the other hand, these drugs are really good and will cure a lot of people and we’re a wealthy country and we can afford it,” he says.
Gren says he’s being forced to take early retirement because he’s becoming too tired to turn up for work every day and too foggy to concentrate on the work that needs to be done.
“I’m not angry…but talk to me again after the PBAC meeting next month and I might be angry if they again refuse to subsidise this new treatment. Obviously, there are no guarantees,” he says.
It’s important for HCV patients to stay positive while waiting for the treatment to become available, advises Dr Levy.
“The people who are infected must try to remember that this is the beginning of a new dawn and it’s fantastic. This is a crazy situation but we’ll get there.”
Graham Osborne is a freelance journalist who has written for The Sydney Morning Herald, SMH, AAP, 2GB, Media Monitors).
Interested in signing the petition to the Minister for Health to take urgent action to expedite the approval of new drugs for hepatitis C, including sofosbuvir?
Like what we do at The AIMN?
You’ll like it even more knowing that your donation will help us to keep up the good fight.
Chuck in a few bucks and see just how far it goes!
Your contribution to help with the running costs of this site will be gratefully accepted.
You can donate through PayPal or credit card via the button below, or donate via bank transfer: BSB: 062500; A/c no: 10495969










11 comments
Login here Register here-
babyjewels10 -
Phi -
Annie B -
Jexpat -
Gary -
Glenn -
Audioio -
CMMC -
Kido -
Sir ScotchMistery -
Annie B
Return to home pageDisgraceful. Let’s share this all over social media.
The truly ugly side of predatory capitalism – at its most brutal. Dangle the cure in front of the afflicted and then let 95% of them die for want of the obscene wealth needed to fill the coffers of the corporate piranha.
How fitting it would be if a global research team could find a pill to cure such greed – it would need to be be administered by force to the likes of Gilead Sciences who epitomise the amoral nadir of predatory capitalism.
Very well said Phi ….
Agree wholeheartedly. ……
and babyjewels – yes it should be spread as far and wide as possible.
Phi:
Wait until you see what Obama & Co. have in store for us with the TPP.
There is a point beyond which the taxpayer cannot be expected to fund extreme profiteering by greedy drug companies. The petition should be directed to the profiteers.
As Jexpat says, wait until you see the mess Abbott and his cronies have got us into with the TPP. Why do you think they won’t let us see the treaty?
remember how Howard was going to sacrifice the PBS on the alter of FTA with USA, and only backed down when Beezley raised the alarm bells with the Australian public. I’m sure Abbott has in his sights to fix that “mistake”.
It would be interesting to know the history of its development. Medicines du Monde is challenging the patent in Europe on the grounds that “the molecule itself is not sufficiently innovative to warrant a patent”.
http://www.medecinsdumonde.org/gb/Doctors-of-the-World-Medecins-du-Monde-is-filing-a-patent-challenge-to-Gilead-s-patent-on-sofosbuvir-at-the-European-Patent-Office
How much of the R&D was done by Gilead and how much of its origins lie in publicly funded research institutions? Predatory capitalism indeed.
I have Hep C, God knows how, only found out after a blood test last year.
Sofosbuvir is what I was hoping for, the hospital can only provide the regular treatment – something like chemotherapy whereby you take handfuls of pills every day and your hair falls out and you just get sick as a dog.
I said thanks, but no thanks. They were planning Sofosbuvir treatment for the near future so I said please contact me when it starts.
Its not going to start, so I prepare for ‘life with a Use-By date’.
Well said Phi. In Australia the Blood Bank’s DID NOT MANDATORILY SCREEN for HEP C until around 1990, therefore the Australian Government should be held accountable with regard to putting this 12 week life-saving treatment on PBS. The number of people with Hep C would be far more and far wider than the stats provided above because there would be many many people walking around carrying this Virus unknowingly, i.e. unless you specifically requested for the Hep C test in a routine blood test. It is always stigmatized in society to only be contracted via I.V. drug use but this is far from fact. If prior to 1990 you had medical procedures, especially blood transfusion’s, plus prior and beyond 1990 tattoo’s, ear piercing and even manicures then everybody is at risk. I witnessed in an Acrylic Nail Beauty Salon a manicure being performed where the staff member using her manicuring tools cut a ladies cuticle and it was bleeding. The un-gloved staff member wiped it with a cotton ball and put the cotton ball straight onto her work station. She put a Band-Aid on the client and continued on. The next client came up to sit down and I said “Aren’t you going to sterilize your manicurist tool’s, your work-station and yourself” first? She looked at me like “what” !! Hep C has no boundaries and is not isolated to I.V. drug use.
Let’s have some perspective here.
Many people don’t care about or understand the TPP.
This little effort is the one that WILL effect many of us mere mortals. The biggest players in the TPP are the drig cartels such as Gilead, Smith Kline Glaxo and Johnson & Johnson.
If the TPP goes through, the access to medication via the PBS list is GONE.
It doesn’t get any simpler than that.
Sir Scotch M …
Have noticed just recently, two prescriptions printed and signed by the Doctor – ‘ were not necessary ‘ … as the items have been taken off PBS, and are over the counter products – at full price. Being a pensioner, I was very reliant on the smallish amount $6.+ for prescriptions that would otherwise cost perhaps over $50 ( or maybe less ).
I would think the Government has already started to take items off the list. And of course, we won’t be told what is coming off and what stays on. … and apparently the Doctors won’t be advised either. … The chemist is a good honest bloke, and stated quite simply – these meds. were no longer covered.
Oh – they are SUCH a reprehensible lot, this mongrel excuse for a Government.